 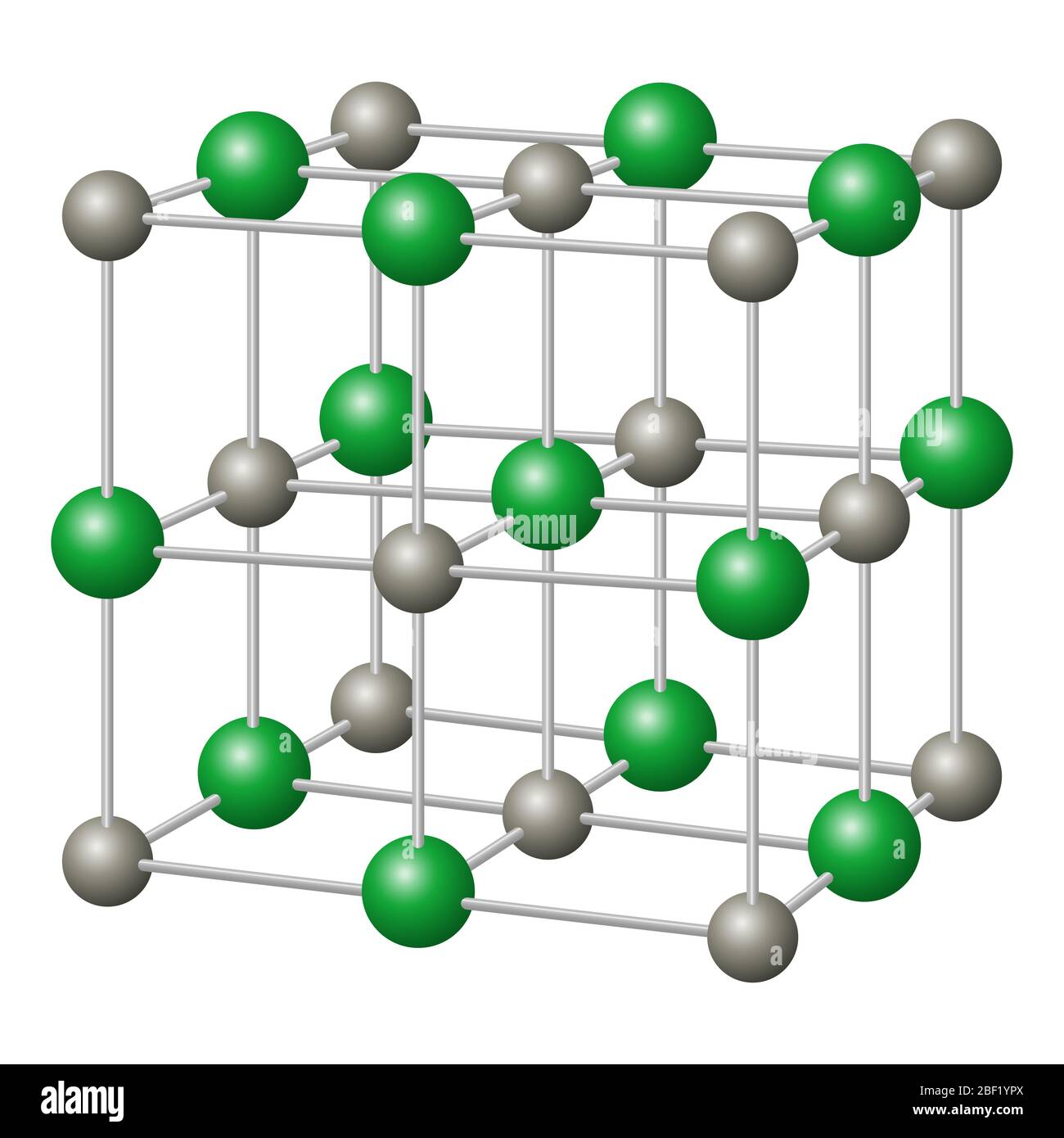
Layered Atom Arrangements in Complex Materials, A Technical Report from Los Alamos National Laboratory, Los Alamos, New Mexico, LA-14205, April 2006. In these structures below, only a single slab of atoms of Na (left) or Cl (right) are shown in the (111) plane. Seen in the (111) plane, one clearly observes that the triangular arrangement of the atoms in each layers. Seen in the (011) plane, one clearly observes the layered structure of the crystal. Ordinary table salt forms a typical cubic close packed. In the system on the right, the crystal is oriented so that the computer monitor is parallel to the (111) plane. This NaCl (sodium chloride) molecular model has 13 grey/sodium atoms & 14 green/chlorine atoms. So that the computer monitor is parallel to the (011) plane. For example, in the system on the left, the crystal is oriented The sodium ions are as close to the chloride ions as possible by fitting into the spaces between them. Compared to sodium ions, chloride ions are significantly larger. Find out how this structure affects the properties of the material, such as melting point, boiling point and conductivity. When rotated, the NaCl crystal structure provides different visualizations of the inherent symmetry. Six chloride ions with opposing charges surround each sodium ion in sodium chloride. Learn how sodium chloride, a giant ionic structure, is formed by the arrangement of positive and negative ions in a three-dimensional pattern. In this structures below, the crystal is oriented so that the computer monitor is parallel to the (001) plane.Ĭrystal structures of arbitrarily large size can be constructed. Below are shown a block of 8 (2x2x2) and 27 (3x3x3) unit cells. The NaCl crystal structure can be generated by periodically reproducing the unit cell. In these structures, Cl is green and Na is light purple. However, visualizing some of the atoms in the neighboring unit cell conveys a better understanding of the crystal structure. Each unit cell contains four Na and four Cl atoms. The following four structures below are equivalent representations of a single unit cell of NaCl. Compounds like this consist of a giant (endlessly repeating) lattice of ions. Sodium chloride is taken as a typical ionic compound. The first atom is located at each lattice point, and the second atom is located half way between lattice points along the face-centered cubic unit cell edge. The structure of a typical ionic solid - sodium chloride. To help show this three-dimensional shape even more accurately, we can rely on space-filling models as well as ball-and-stick models.The sodium chloride structure adopts a face-centered cubic lattice with a two-atom basis or as two interpenetrating face centered cubic lattices. We will discuss the significance of these electrons at the end of this section. The two dots above nitrogen indicate a lone pair of electrons that are not involved in any covalent bond. As with any FCC lattice, there are four atoms of sulfur per unit cell, and the the four zinc atoms are totally contained in the unit cell. However, in the more detailed structural formula on the right, we have a dashed line to indicate that the rightmost hydrogen atom is sitting behind the plane of the screen, while the bold wedge indicates that the center hydrogen is sitting out in front of the plane of the screen. This structure consists essentially of a FCC (CCP) lattice of sulfur atoms (orange) (equivalent to the lattice of chloride ions in NaCl) in which zinc ions (green) occupy half of the tetrahedral sites. 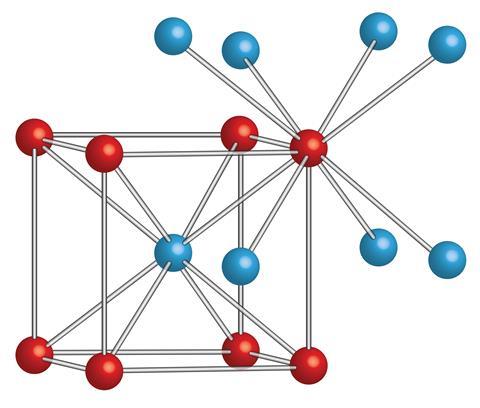
In the structural formula to the left, we are only seeing a two-dimensional approximation of this molecule. Keep in mind, however, that atoms and molecules, just like everything else in the universe, exist in three dimensions-they have length and width, as well as depth. From both of these structural formulas, we can see that the central nitrogen atom is connected to each hydrogen atom by a single covalent bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
